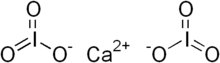Lautarite
Calcium iodate
Chemical compound
Calcium iodate is any of two inorganic compounds with the formula Ca(IO3)2(H2O)x, where x = 0 or 1. Both are colourless salts that occur as the minerals lautarite and bruggenite, respectively. A third mineral form of calcium iodate is dietzeite, a salt containing chromate with the formula Ca2(IO3)2CrO4.[1] These minerals are the most common compounds containing iodate.
This article relies largely or entirely on a single source. (November 2023) |