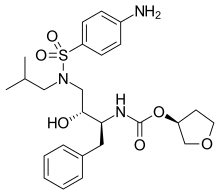
Amprenavir
Amprenavir
Chemical compound
Amprenavir (original brand name Agenerase, GlaxoSmithKline) is a protease inhibitor used to treat HIV infection. It was approved by the Food and Drug Administration on April 15, 1999, for twice-a-day dosing instead of needing to be taken every eight hours. The convenient dosing came at a price, as the dose required is 1,200 mg, delivered in 8 (eight) very large 150 mg gel capsules or 24 (twenty-four) 50 mg gel capsules twice daily.[1]
This article needs additional citations for verification. (December 2009) |
It was patented in 1992 and approved for medical use in 1999.[2] Production of amprenavir was discontinued by the manufacturer on December 31, 2004; a prodrug version (fosamprenavir), is available.